|
9/8/2023 0 Comments Enthalpy vs entropy 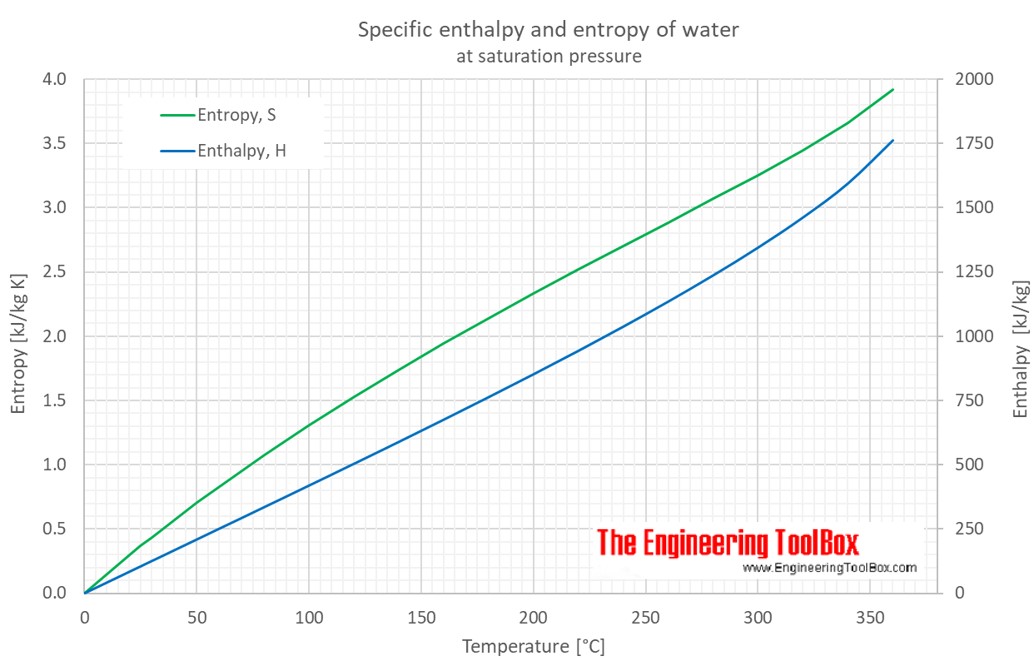
Thus although the free energy always falls when a gas expands or a chemical reaction takes place spontaneously, there need be no compensating increase in energy anywhere else. Free Energy is not energy: A much more serious difficulty with the Gibbs function, particularly in the context of chemistry, is that although G has the units of energy (joules, or in its intensive form, J mol –1), it lacks one of the most important attributes of energy in that it is not conserved.This is most commonly in the form of electrical work (moving electric charge through a potential difference), but other forms of work (osmotic work, increase in surface area) are also possible. The Mollier enthalpyentropy diagram for water and steam. By "useful", we mean work other than that which is associated with the expansion of the system. The “free” part of the older name reflects the steam-engine origins of thermodynamics with its interest in converting heat into work: ΔG is the maximum amount of energy which can be “freed” from the system to perform useful work. 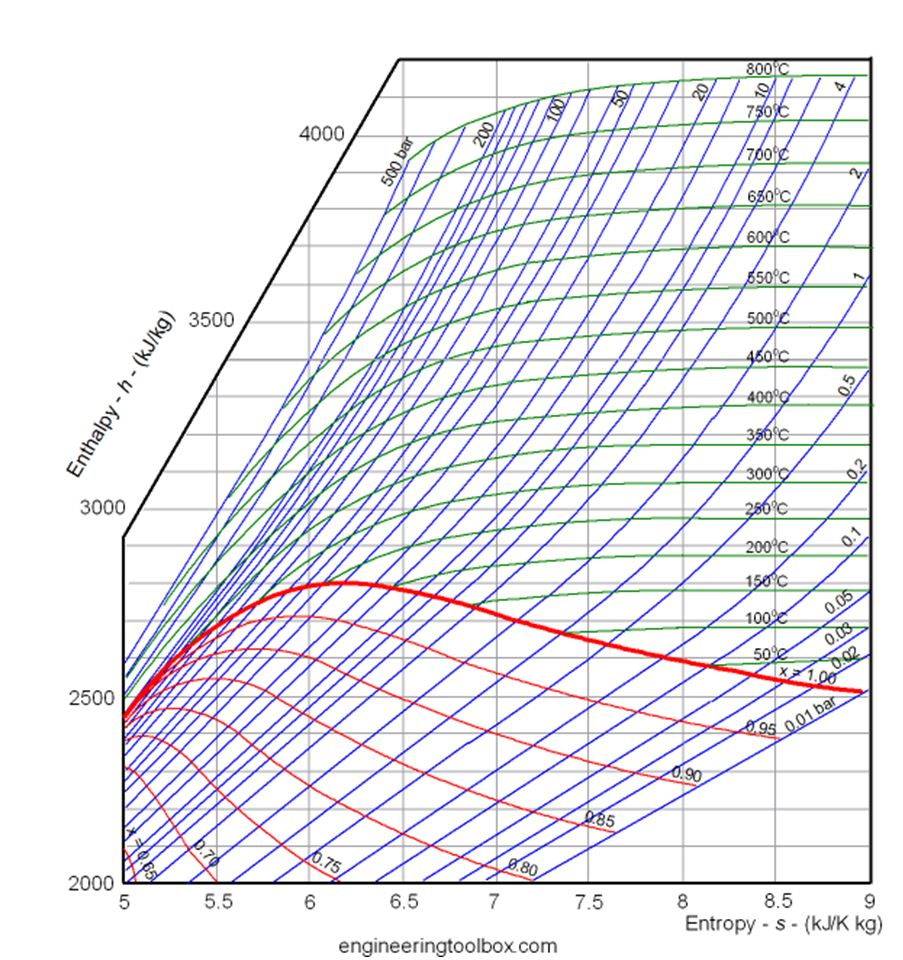 
Gibb’s free energy provides the relationship between enthalpy and entropy. The main difference between enthalpy and entropy is that enthalpy is a measure of the total energy of a system that includes the effects of pressure and. Entropy is a change in enthalpy per unit temperature. Free Energy is not necessarily "free": The appellation “free energy” for G has led to so much confusion that many scientists now refer to it simply as the Gibbs energy. What is the relationship between enthalpy and entropy Enthalpy is the amount of energy released or absorbed in a system whereas, entropy is the measure of disorderness in a system. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
